Basics of Heat Treatment for Metallic Material (Part-1)
Scope:
In this article fundamentals of heat treatment will be discussed briefly mainly related to the “base material” of welded construction (metallic material) meant for industrial application.
In Part-0, we had discussed the basics from a theory point of view generally taught in engineering colleges (Click here to read Part – 0).
In the present article i.e Part – 1, we will discuss heat treatment from the industrial application point of view mainly for the materials to be joined by welding (Base materials).
Importance of Heat Treatment:
Properties of steel can be changed considerably through heat treatment in its solid-state, at a specified chemical composition. The reason for this is the capability of the great majority of iron-carbon alloys to be transformed into polymorphous lattice formations.
Need for Heat Treatment in welded Structure (to name a few) :
- To relieve the residual stresses (ferritic steels).
- To dissolve the carbides (chromium carbides) to avoid sensitization (intergranular corrosion) especially in austenitic steel.
- To make the microstructure of the steel especially low and high alloy steel (Cr-Mo, 13Cr-4Ni, etc.) suitable for service (tempered martensite).
Heat Treatment based on when it is performed:
- Intermediate Heat Treatment: During cold forming or for high restraint joints in Cr-Mo Steel (Nozzle to Shell/Dish end joint).
- Post Weld Heat Treatment: Performed immediately after or after allowing the weldment to cool to room temperature with an aim as mentioned in the above points.
Classification of Heat Treatment Process (Base Material):
- Annealing
- Hardening and
- Quenching/Tempering.
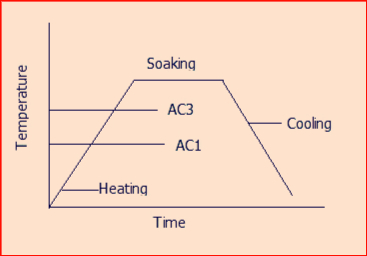
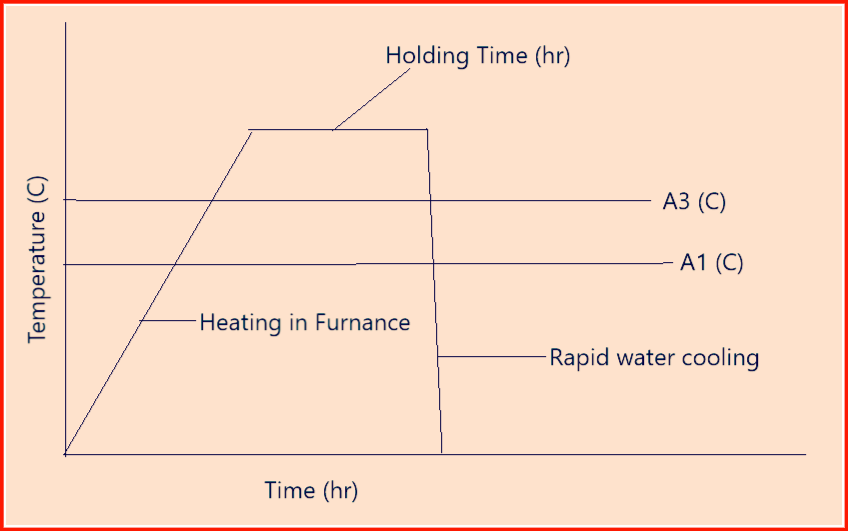
Referring to figure 1 of Article-0 below features the characteristic temperature ranges for important heat treatment processes in the Fe-FeC system. It is evident that the heat treatment temperature is often dependent on the carbon content.
Where;
A1= The temperature at which austenite beings to form during heating,
A3= The temperature at which the transformation of ferrite to austenite is complete during heating.
Annealing: It’s a general term denoting a thermal treatment that consists of heating at a specific rate, holding at a specific temperature for a finite period of time and cooling at a specified rate primarily to soften the metallic materials.
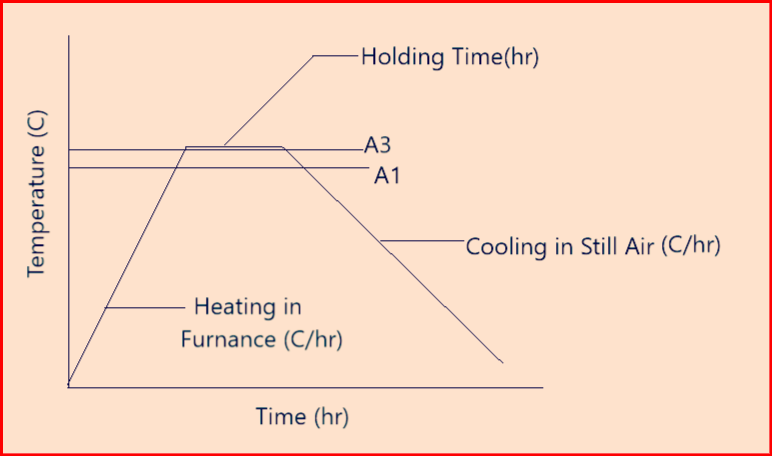
Above figure-2 shows an example of Annealing (Homogenizing)
Here:
A1 = the temperature at which austenite beings to form during heating
A3 = the temperature at which the transformation of ferrite to austenite is completed during heating.
There are many types of annealing depending on the changes in material properties desired namely:
- Achieving uniform microstructure:
- Diffusion annealing
- Normal annealing
- To enhance workability
- Coarse grain annealing
- Soft annealing
- Recrystallization annealing
- Crystal recovery annealing
- Minimizing the stresses
- Stress relief annealing
- Stress-relieving treatment
- Microstructural changes
- Tempering
- Special Cases
- Controlled Cooling
- Quenching
- Perlitic annealing
- Bainitic microstructure transformation
- Controlled Cooling
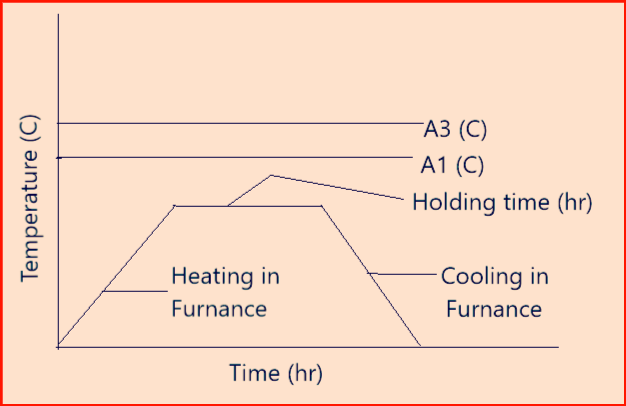
Normalizing: Heating to 30 to 50°K above A3 (austenising) temperature in case of hypoeutectoid (carbon steel) steel. The soaking time at hold temperature will be such that the whole job is heated uniformly to the normalizing temperature. After that, it will be cooled in the resting air.
The aim of normalizing is thus also to achieve an even microstructure with fine lamellar pearlite.
- Above figure-3 represents a normalizing cycle.
- Normalizing is recommended in the following cases:
- In case of coarse grain.
- In the case of solidification microstructure, eg weld joint or cast steel
- In the case of steels embrittled due to aging and
- In the case of all constructional steels with an insufficient toughness or too low a yield point.
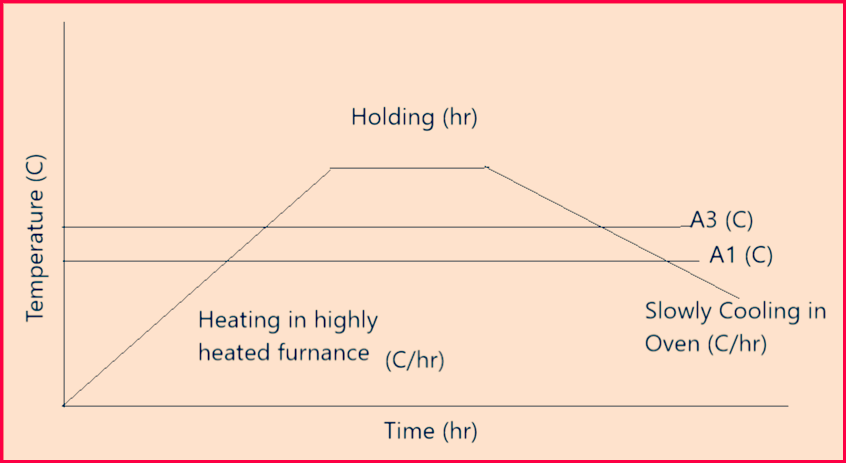
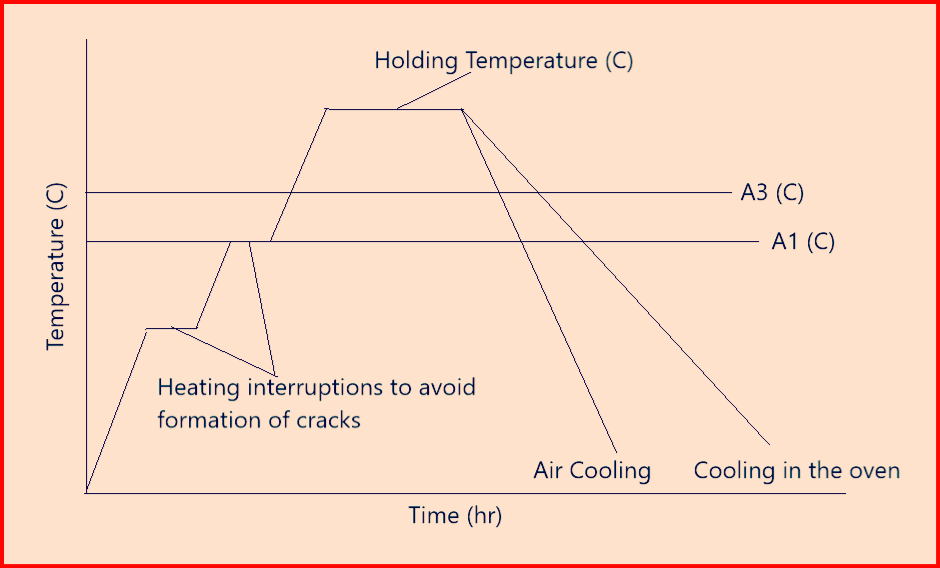
Stress Relief annealing: Heating to and holding at annealing below A1 with subsequent slow cooling, so that internal stresses are reduced without significant changes to the other properties
- Above figure-4 represents a stress relief annealing cycle.
- Internal stresses can be generated due to uneven heating and cooling of the material e.g. during welding, soldering, solidification, or quenching but also due to cold forming. These stresses often cause distortion or lead to the formation of cracks in the component.
Coarse grain annealing: it is carried out in the temperature range of 950° and 1100° C (above A3) with the hold time of about 1hr to 4 hr (Prolonged period of time) and cooling slowly to achieve coarse grain structure.
The aim of coarse grain formation is to produce a coarse grain with low hardness and toughness. This type of microstructure is advantageous for milling, since the chips produced are small, thus leading to lower cutting forces.
- Above figure-5 represents a coarse grain annealing cycle.
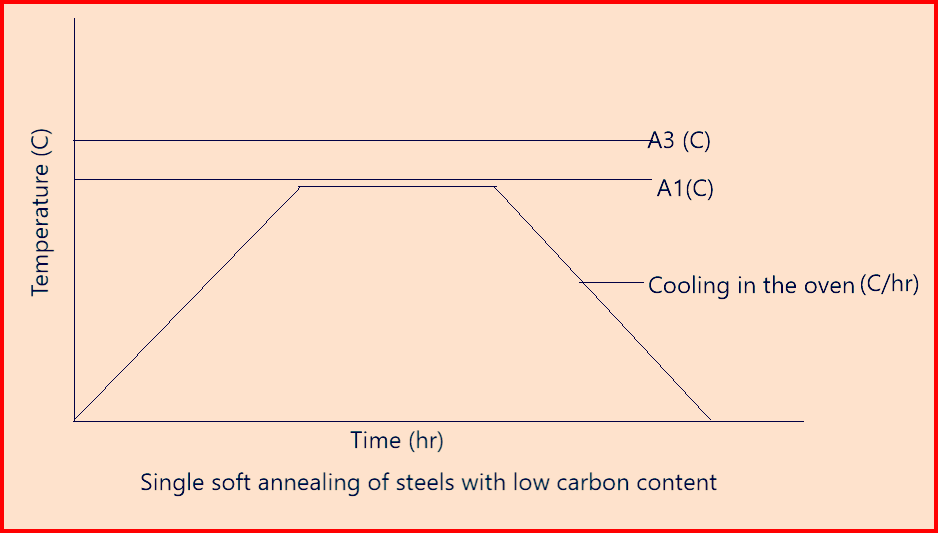
Soft annealing (annealing on globular cementite): It is meant to reduce the hardness of the material to a defined value in which annealing is performed at temperature closely below or just above A1or held around A1 with subsequent slow cooling.
- The aim of soft annealing is to obtain a soft microstructure suitable for further processing, such as milling or cold deformation.
- The optimum condition of the microstructure consists of homogeneously distributed, fine-grained, globular carbides embedded in a ferritic matrix and such a microstructure can be split, bent, compressed, flanged, extruded or twisted with ease.
- Below figure-6 represents the soft annealing cycle of low carbon steel.
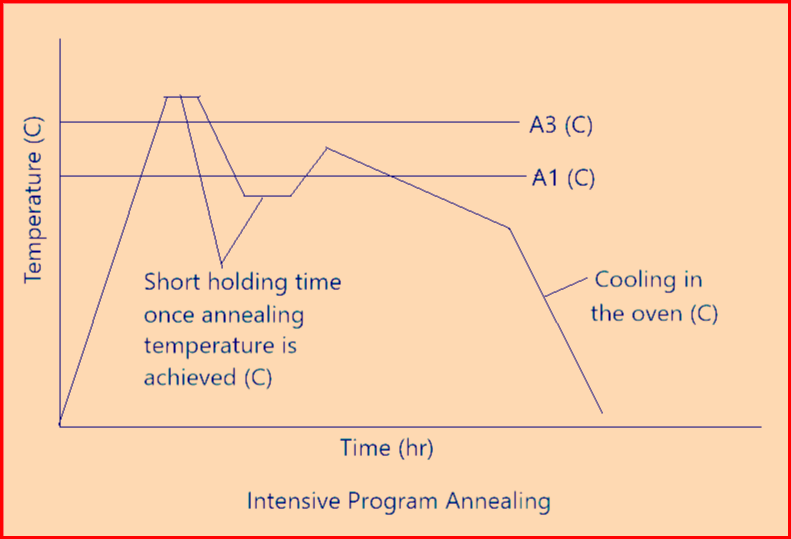
- The above figure-7 represents heat treatment (2nd picture) which is mainly used in steels with carbon content above 0.5%, for example, with roller bearing steels.
Diffusion annealing: Annealing in the temperature range of 1050°C to 1250°C, with a prolonged hold time of around 50hr, in order to balance local differences in the chemical composition due to segregation by diffusion.
- Due to high temperatures during diffusion annealing a formation of coarse grains is unavoidable. Coarse grain can be removed by normalizing.
- Below figure-8 represents a diffusion annealing cycle.
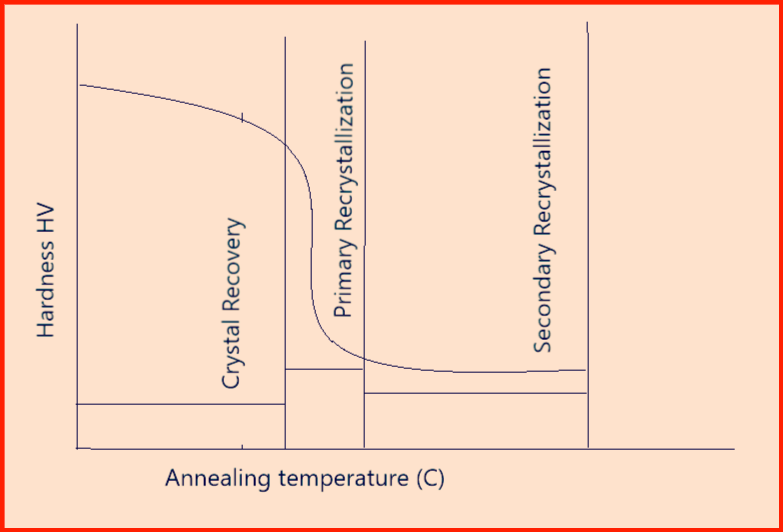
Recrystallization annealing: Heat treatment to realize new grain formation in a cold-formed workpiece through nucleation and growth without a phase change.
- During cold forming as the hardness increases which reduces the formability and for this reason, cold deformation cannot be carried out indefinitely and hence after certain deformation recrystallization annealing in order to restore the original material properties.
- Min recrystallization temperature = 0.4 times absolute melting temperature.
- Three Stages involved are as mentioned below:
- Crystal Recovery: It involves rectifying dimensional lattice structure imperfections and rearranging dislocations.
- Primary recrystallization: It leads to complete grain reforming.
- Secondary recrystallization: Above the recrystallization temperature, the small grains formed by primary recrystallization combine to form a small number of large grains.
- Above figure-9 represents a recrystallization annealing cycle.
Solution annealing: The main aim of solution annealing is to bring and hold separated microstructures in solution (homogenizing).
- This is particularly desirable with austenitic Cr-Ni steels (above 1000 C) because it enables the required corrosion resistance to be achieved.
- The purpose is to obtain a standardized austenite crystal deprived of precipitations by rapidly cooling in chilled/room temperature water.
- Above figure-10 represents a solution annealing cycle.
(Note: The purpose of this article is to give a general guideline to the readers, and it shall not be considered as a substitute for code. For full terms and conditions please read relevant code and its clauses).
This article is written by;
MR. SANDEEP SINGH PARMAR
(Ex. GE, ISGEC & ESSAR)
Email: sandeepparmar99@yahoo.com
IWE (IN/IWE/41700026); B Tech (Mechanical); AMIIW (Welding Technology)
ISO 9001:2008 Internal Auditor; ISO 9001:2015 Lead Auditor ;
NDE L-II (UT, LPT, MPI, RT); Lean Six Sigma Green Belt;
MWeldl IEng; MIE C Eng(Ind) ; M.I.Inst.W ; LM IIM